
With fewer 90° LP–BP repulsions, we can predict that the structure with the lone pair of electrons in the equatorial position is more stable than the one with the lone pair in the axial position. We use Lewis symbols to describe valence electron configurations of atoms and monatomic ions. If we place it in the axial position, we have two 90° LP–BP repulsions at 90°. Identify the Central Atom Recognize that carbon will be the central atom. Is the lewis structure of NCl3 the same as NF3 and. You can see the 7 valence electrons present in the chlorine atom as shown in the above image. 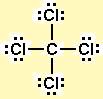
This model assumes that electron pairs will arrange themselves to minimize repulsion effects from one another. according to the VSEPR chart, the electron geometry of NCl3 is tetrahedral while molecular geometry is trigonal pyramidal. The valence shell electron pair repulsion (VSEPR) theory is a model used to predict 3-D molecular geometry based on the number of valence shell electron bond pairs among the atoms in a molecule or ion. It has a melting point of 40☌ and a boiling point of 71☌. The equation for dipole moment is as follows. Dipole moment is equal to the product of the partial charge and the distance. The net dipole is the measurable, which is called the dipole moment. However, because the axial and equatorial positions are not chemically equivalent, where do we place the lone pair? If we place the lone pair in the equatorial position, we have three LP–BP repulsions at 90°. It is not soluble in water but soluble in benzene, PCl3, CCl4, etc. Using the cross bow arrow shown below we can show that it has a net dipole. Well start by looking at the valence electrons. 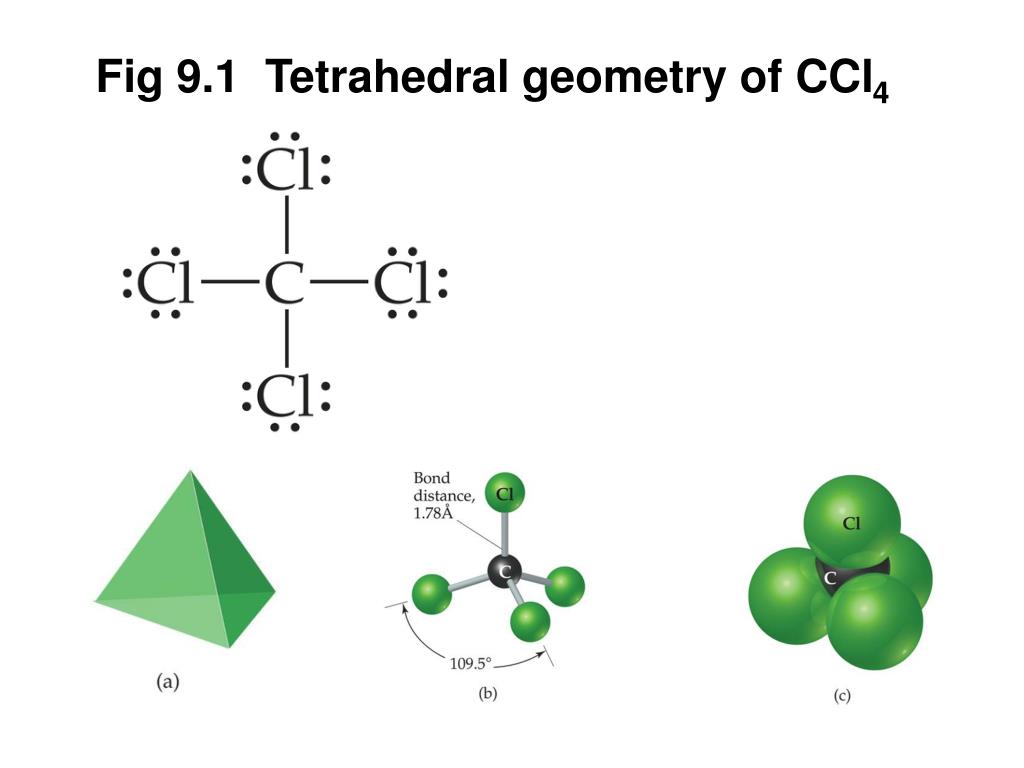
We designate SF 4 as AX 4E it has a total of five electron pairs. Lets do the Lewis structure for CCl4, Carbon Tetrachloride, sometimes just called Carbon Tet.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
